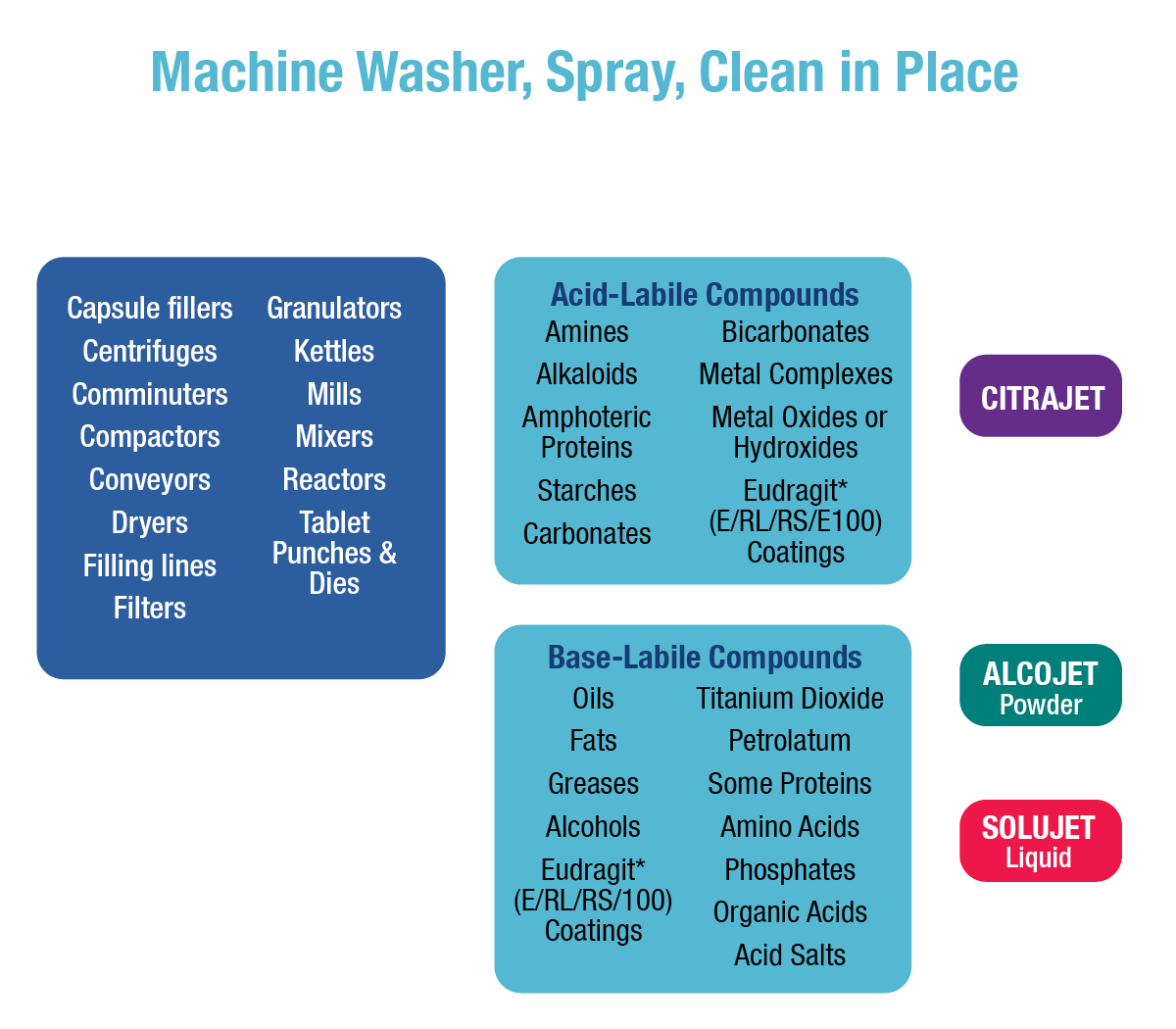
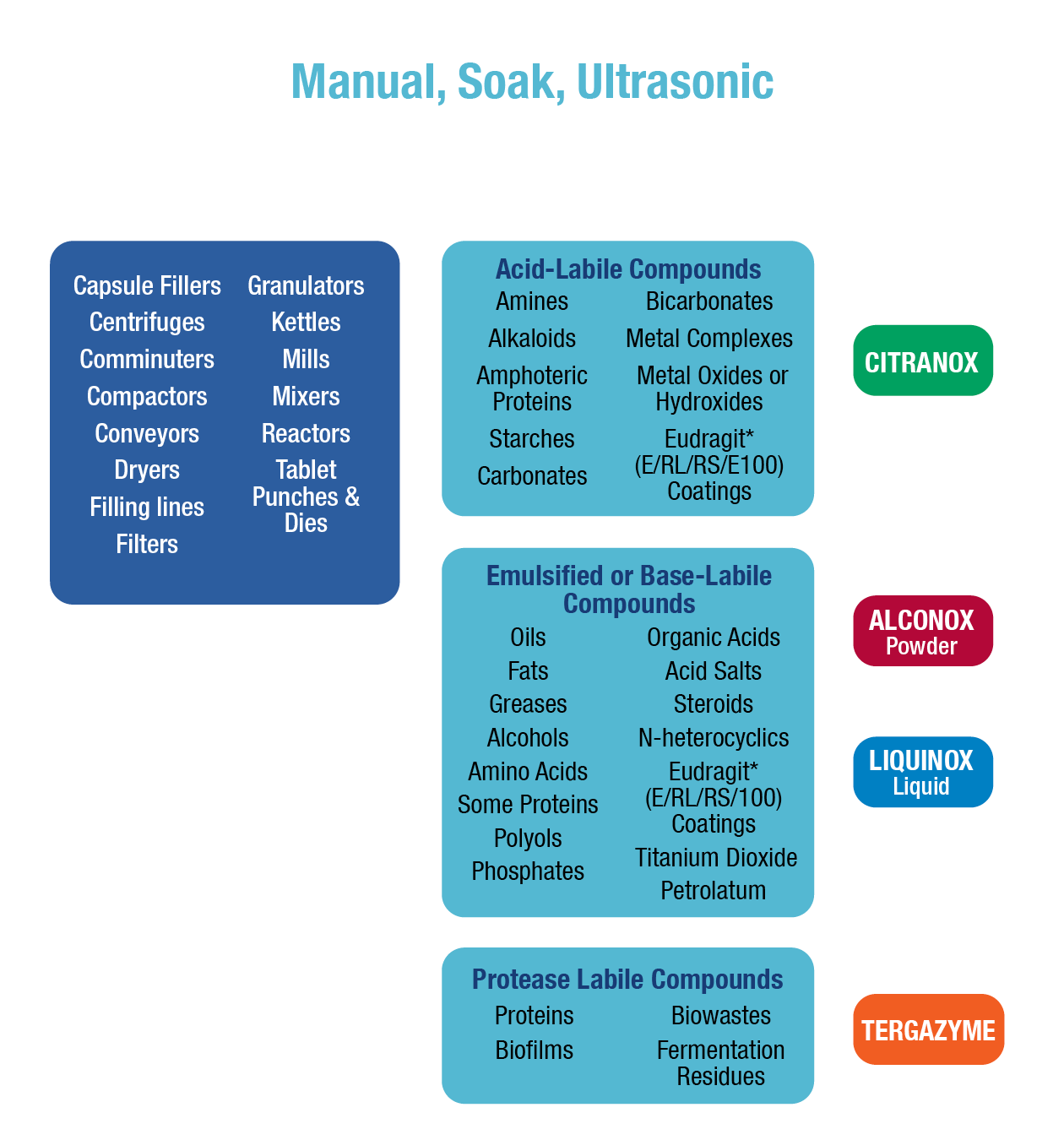
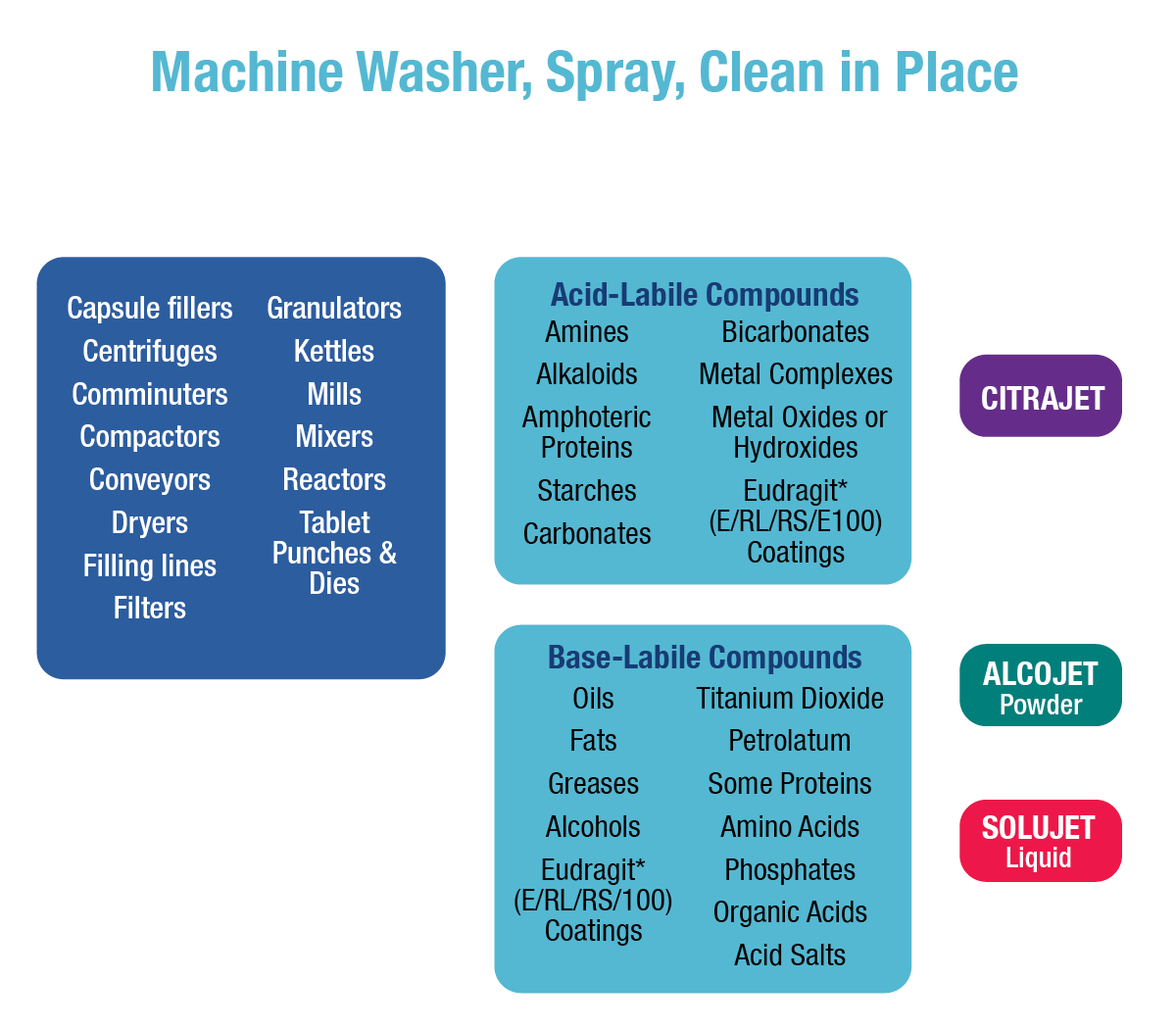
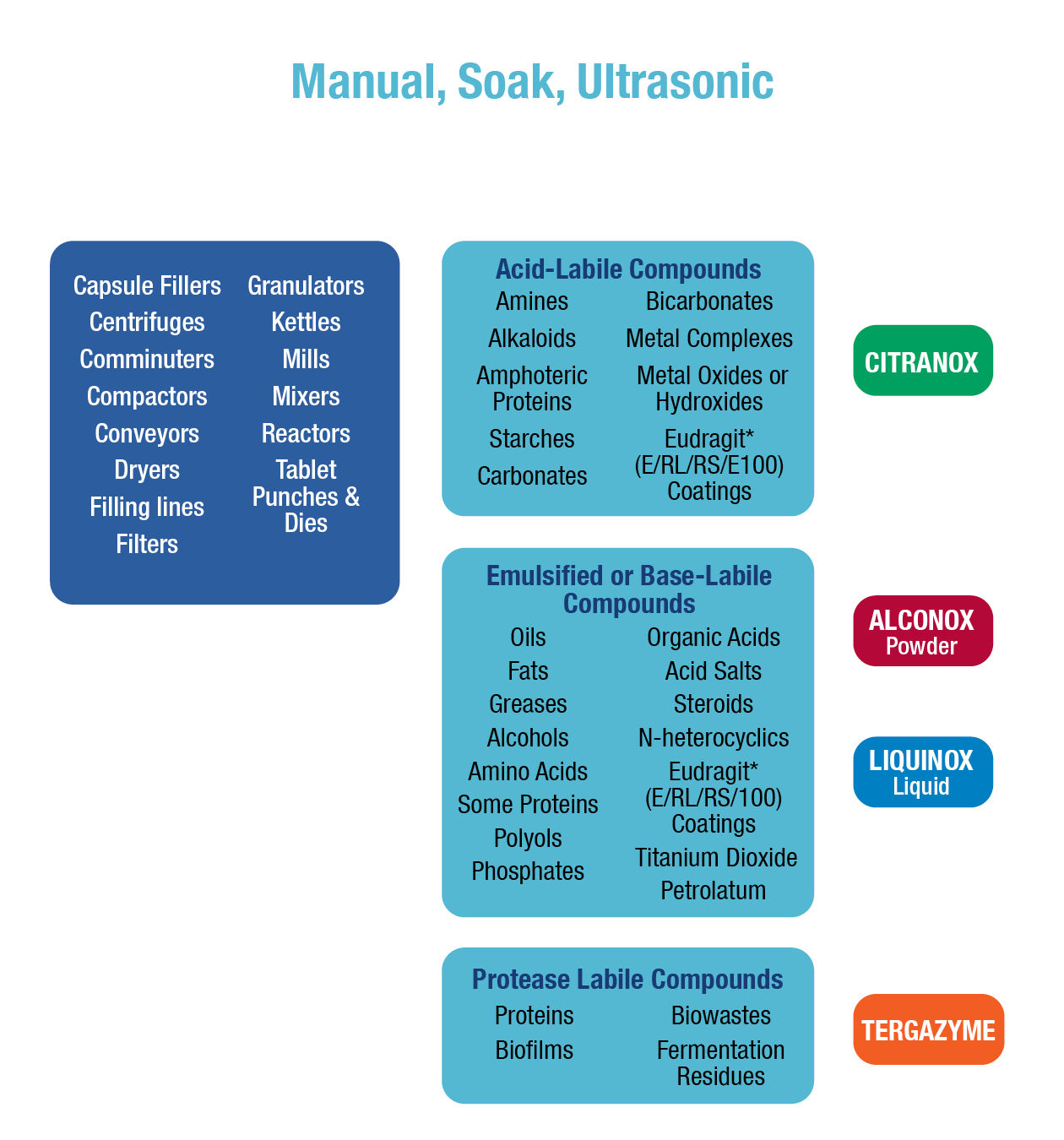
Pharmaceutical equipment includes everything from bench-scale laboratory apparatus used for trials to full-scale bulk manufacturing equipment. Regardless of scale or complexity, all pharmaceutical equipment must meet government standards for cleanliness. Residues found in pharmaceutical cleaning range from easy-to-clean water-soluble excipients to difficult-to-clean petrolatum/metal oxide mixtures. To simplify regulatory compliance and reduce the probability of using the wrong detergent, it is desirable to use as few cleaners as possible to remove the entire range of residues encountered. It also is desirable for these cleaners to work in a wide range of cleaning procedures, including manual, soak, and ultrasonic cleaning, as well as clean-in-place (CIP) spray systems. Alconox, LLC meets these pharmaceutical cleaning requirements and provides support for pharmaceutical cleaning validation, lot number traceability, biocompatibility, toxicity data, shelf-life testing, and residue detection methods (analytical methods).